AHEAD-Flow Platform
AHEAD delivers on-demand insights by augmenting human visual interpretation with AI and machine learning. As the software intelligence layer between machine generated cell-level details and humans, the AHEAD platform offers AI/ML enhanced analytic productivity: 10X-20X faster and reproducible results at 3-6X lower cost for vaccine and drug development, cell therapy QA/QC and clinical diagnosis and monitoring.
Low cost, on-demand insights, and higher productivity enable AHEAD to relieve bottlenecks and enable global availability.

Anbogen Therapeutics
Anbogen Therapeutics is a clinical stage precision oncology company developing first/best-in-class personalized therapeutics for the treatment of solid tumors and blood cancers. We aim to bridge the gap between discovery candidates and proof-of-concept indications for unmet needs in clinical studies. We are advancing a pipeline of precision medicines for the treatment of solid tumors and blood cancers. Currently with three assets including ABT-101 and ABT-301 in clinical development.
product introduction
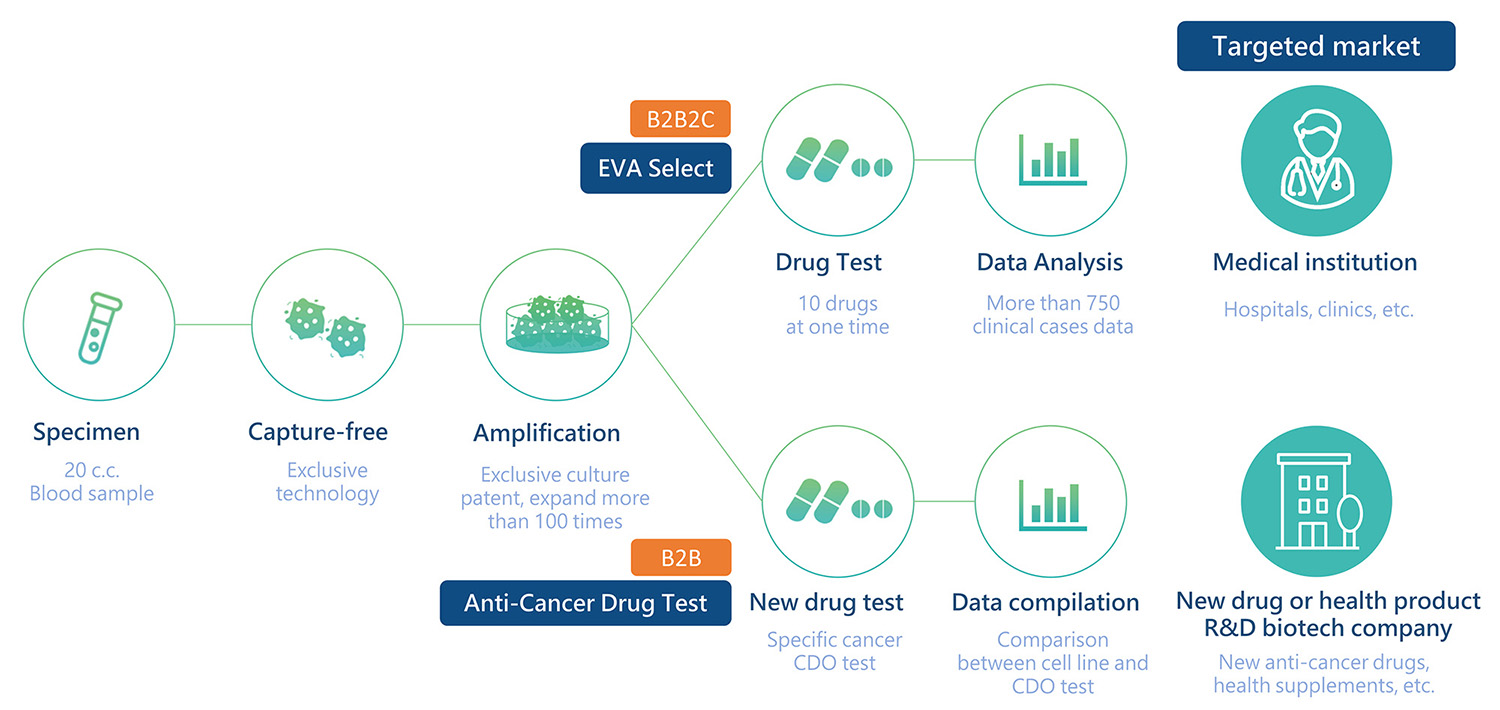
With only 20 ml of blood samples, our proprietary platform E.V.A. can proliferate and amplify circulating tumor cells (CTCs) directly for many applications in cancer research, such as drug selection, cell therapy, cancer vaccine and new drug development.
DeepCT volume
Deep01 Limited
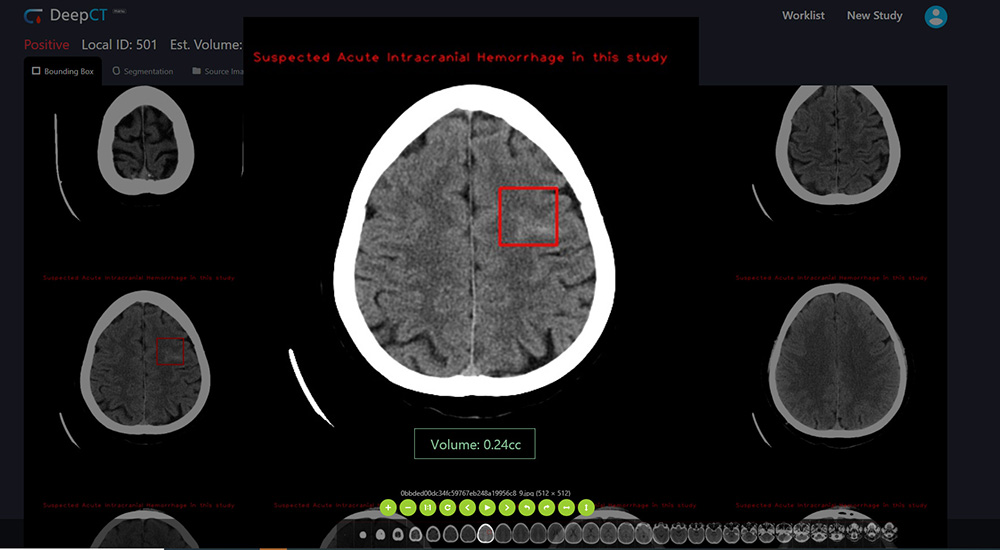
Deep01 is the developer of DeepCT - world-leading AI-powered brain CT abnormalities detection and quantification system. It detects brain bleeds, fractures, midline shift, and calculates ICH Score automatically.
Our AI makes doctors more efficiently in the medical process:
For Neurosurgeons - help to evaluate patients
For Emergency Physicians - help to triage patients
For Radiologists - help to generate reports
Product
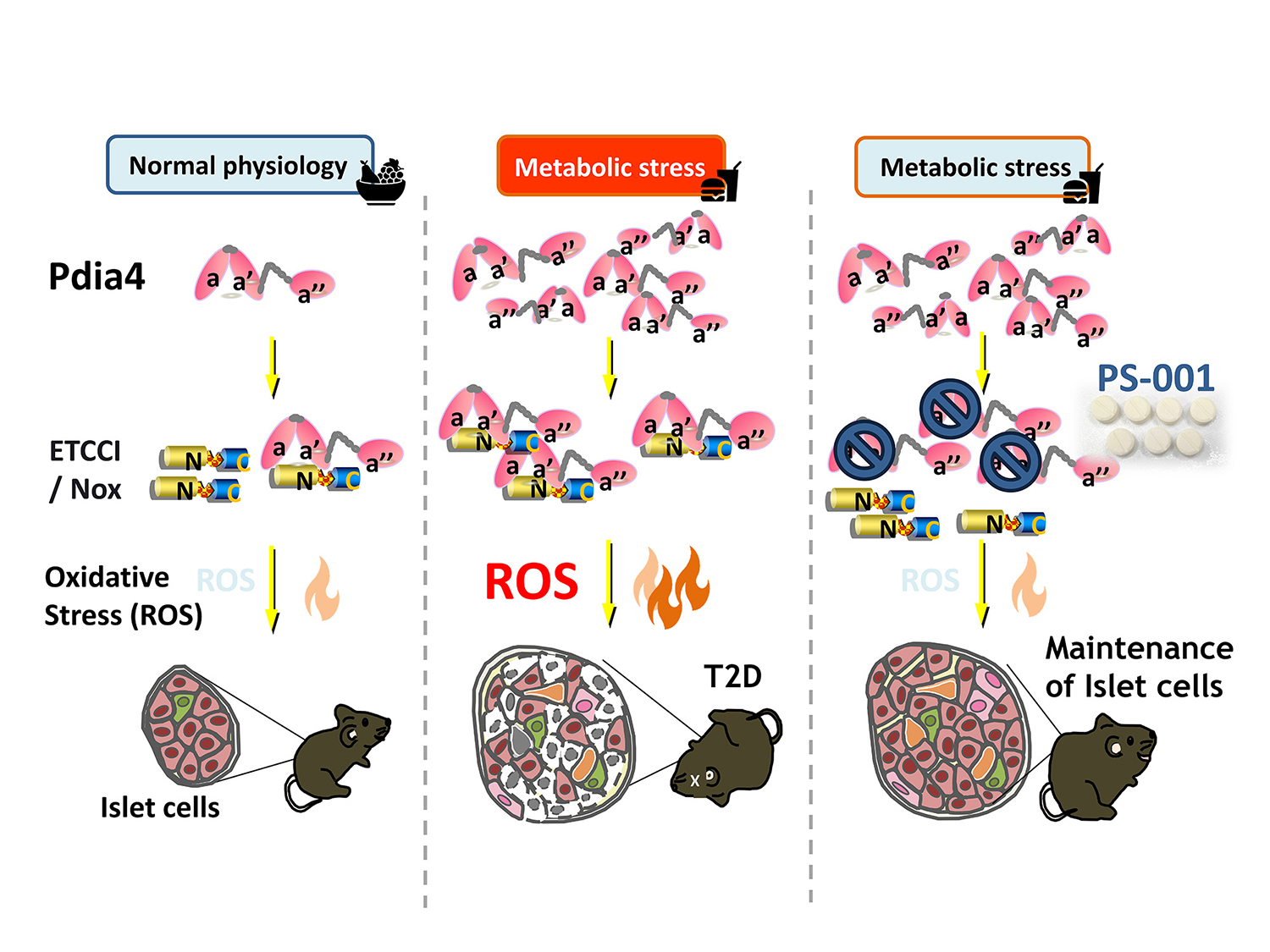
Metabolic stress up-regulates Pdia4 in ß-cells, leading to exuberant oxidative stress and, in turn, ß-cell failure and diabetes. PS-001, a first-in-class Pdia4 inhibitor, can prevent ß-cell failure and reverse diabetes.
CancerFree Biotech Ltd.
CancerFree Biotech focuses on the use of Circulating Tumor Cells-Derived Organoid (CDO) culture system for a personalized analysis. We can rapidly amplify CTCs and develop an optimized anticancer strategy from the currently available anticancer drugs through low-risk and convenient liquid biopsy and biomimetic organoid culture system.
FaceHeart Corp.
FaceHeart Corp. was born out of National Yang Ming Chiao Tung University’s Electrical and Computer Engineering department in 2018 under Dr. Bing-Fei Wu, IEEE Fellow. FaceHeart is on a mission to improve human health by transforming the healthcare, home automation, and insurance industries through its proprietary video-based rPPG technology. The solution is packed into FH Vitals™ SDK, allowing seamless integrations into a variety of settings to remotely measure key vital signs.

ImmunAdd Inc.
Founded in 2021, ImmunAdd, Inc., is a privately held biopharmaceutical company on a mission to improve health through advances in adjuvant design and formulation. We are developing next generation saponin adjuvants and using them as the basis for new combination adjuvants and adjuvanted vaccines.
Our lead adjuvant, IA05, is a rationally designed analogue of the saponin adjuvant QS-21. IA05 elicits immune responses comparable to or better than QS-21 and is better tolerated and more stable.
With IA05 as the foundation, ImmunAdd is designing adjuvant platforms able to deliver a specific immune response.

PrecisemAb Biotech Co., Ltd.
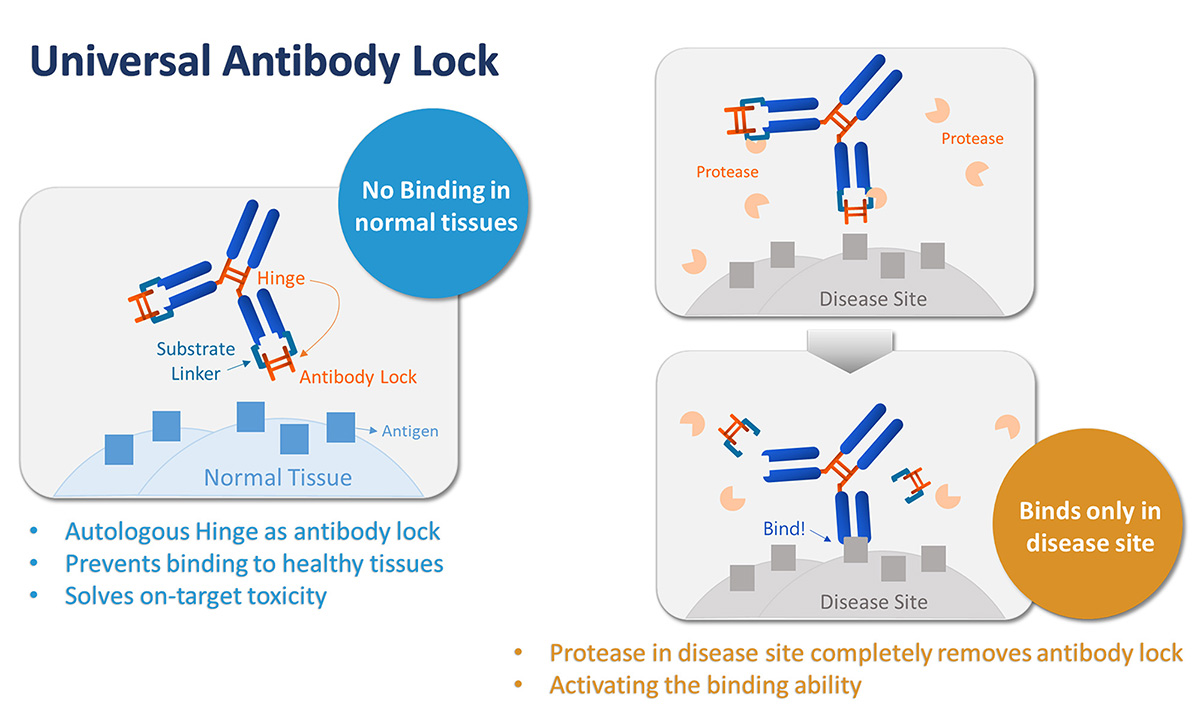
We are PrecisemAb, a company that is committed to blocking side effects caused by antibody therapy. Our disease-directed Universal Antibody Lock is an autologous masking structure on the binding site of an antibody that allows antibody drugs to take effect only in diseased areas. This makes antibody treatment extraordinarily safe.
Compared to competitors, our Antibody Lock
1) is the 1st antibody lock that uses the antibody's own structure as a masking domain.
2) has higher masking ability and can fully activate the antibody when in the diseased areas.
3) has low immunogenicity and non-anti-idiotypic antibody binding, which prolongs serum half-life and leads to higher therapeutic efficacy.
4) takes only 3 months for Lock-design and function test.
We have successfully applied the technology to autoimmune diseases (TNFα and IL-1b) and immuno-oncology therapy (CTLA4, PD-1, 4-1BB). Potential application to ADC drugs is now under lead optimization. Through providing a customized lock-design service and co-developing lock-antibody drugs with pharma, PrecisemAb is dedicated to creating the safest antibody therapy for patients.

Biosensor

APrevent Medical
APrevent is a medical device company, founded by a team of international clinically experienced physicians and engineers. APrevent is dedicated to providing minimally invasive and innovative laryngeal implant and Software-as-a-Service (SaaS) technologies for voice & speech disorders, which are patient-orientated, physician friendly, and improve the quality of life for patients around the world. APrevent Intelligible speak, AiSpeak, is an innovative solution using big data and AI machine learning technology providing patients with motor speech disorders to communicate with their own speech, which recognizes and converts dysarthric speech into intelligible speech in real time. It can also diagnose and monitor conditions of a neurological disease based on the changes and variations of acoustic characteristics from patients. To help dysarthric patients regain normal communication capability and life quality, APrevent is developing several innovative solutions based on AiSpeak technology platform, including Portal and Messenger. AiSpeak opens a door to a complete new dimension of user experience for dysarthric patients with high potential value.
FaceHeart Corp.
FaceHeart Corp. was born out of National Yang Ming Chiao Tung University’s Electrical and Computer Engineering department in 2018 under Dr. Bing-Fei Wu, IEEE Fellow. FaceHeart is on a mission to improve human health by transforming the healthcare, home automation, and insurance industries through its proprietary video-based rPPG technology. The solution is packed into FH Vitals™ SDK, allowing seamless integrations into a variety of settings to remotely measure key vital signs.
Molsentech
Molsentech develops a 100% electrical-based and label-free disease detection platform powered by the Bio-FET technology that offers real-time and ultrahigh-sensitive detections.
Current detection platforms like PCR and ELISA are optical-based and there are complicated pretreatments needed to be done before the running real detections. Most of the time, their bulky equipment leads to high running and maintenance costs. To fix these pain points, we take advantage of the electrical-based Bio-FET technology that don’t require bio-labeling or amplification in pretreatments, so we can save a lot of time in the whole process. To be more specific, our analyzer can measure the immediate change in signals coming from the interactions between detection targets and bio probes we modify on the surface of biosensors to generate real-time results.
Our successful demo was COVID-19 RNA rapid test. Our testing time is as fast as antigen rapid tests, but we provide PCR-level accurate detections. Importantly, we just need about 3 months to develop a different biosensor assay for a new application and have started developing customized biosensors for biotech companies, which is our core B2B business model. This way, we could grow up by becoming a biosensor design house doing licensing and ODM so that we can expand our customer base and make a greater impact.
Our R&D members are from Academia Sinica and we have been on the BioFET research field for more than 2 decades. In addition to publishing papers with top SCI journals, in 2016, Molsentech signed a Joint-Development-Agreement with TSMC to develop Bio-FET biosensors together, the fact not only helped us save huge capital but also recognized our capability of facilitating this technology in the business world.
All in all, Molsentech has leveraged Taiwan’s semiconductor industry to make a promising technology ready to commercialize. Our vision is to become an important contributing factor to preventive healthcare & precise medicine. Molsentech is looking for strategic partners and investors, especially in the U.S., to successfully reach this goal with us.
ABT-101
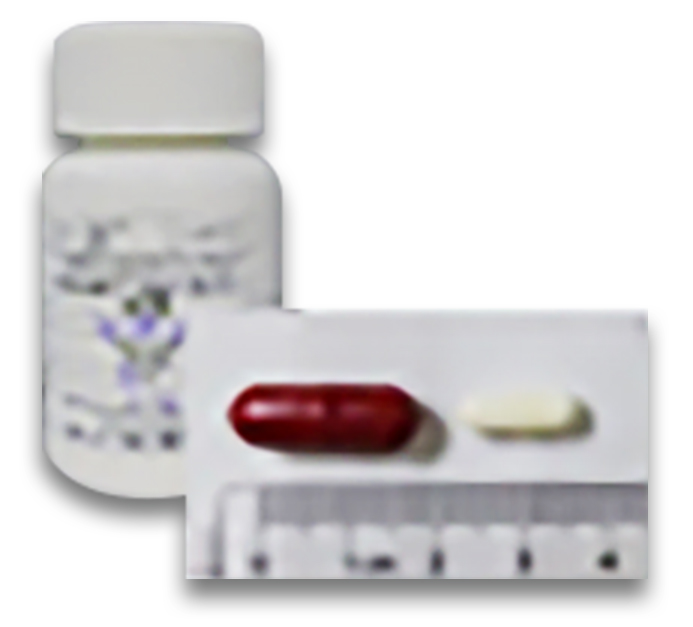
ABT-101, A small molecule drug targeting HER2 exon 20 insertion.
ImmunAdd Inc.
Founded in 2021, ImmunAdd, Inc., is a privately held biopharmaceutical company on a mission to improve health through advances in adjuvant design and formulation. We are developing next generation saponin adjuvants and using them as the basis for new combination adjuvants and adjuvanted vaccines.
Our lead adjuvant, IA05, is a rationally designed analogue of the saponin adjuvant QS-21. IA05 elicits immune responses comparable to or better than QS-21 and is better tolerated and more stable.
With IA05 as the foundation, ImmunAdd is designing adjuvant platforms able to deliver a specific immune response.

AHEAD Medicine
AHEAD Medicine unlocks the potential of flow cytometry and other high dimension data sources.
Increasingly precise lab equipment has begun to outstrip the capabilities of highly trained physicians and researchers, leading to delays, discordance, and excess costs. AHEAD uses machine learning and artificial intelligence to aggregate data across sites, provide deeper insights, and deliver faster and more accurate data interpretation all while lower testing costs.
Currently available for RUO, find out how we can help accelerate your research.

AcadeMab Biomedical Inc.,
AcadeMab Biomedical Inc., established in 2020, is a biotechnology company dedicated to discovering and early-stage development of proprietary biopharmaceuticals that applying our antibody development platforms. Our extensive practical experience with the platforms allows us to leverage resources and transform innovation that bringing up impact on disease treatment.

Analyzer-SENEDIA

Calgent Biotechnology Co. Ltd.
Calgent Biotechnology Co. Ltd.
CALGENT, a spin-off company of Taipei Medical University (TMU), was established in 2019 and thrives on applying science and technology to drug development. CALGENT is committed to developing first-in-class drugs for novel therapeutic cancer targets and various treatments for ocular system, inflammatory and immune system related diseases.
One of the CALGENT’s main objectives is to develop exceptional and innovative medicines with the potential to bring positive and profound impacts on improving the life quality for patients with unmet needs.
Vision and Mission
CALGENT primarily focuses on the process from preclinical to clinical proof of concept (PoC) during the drug development value chain. CALGENT primarily targets highly novel projects with new molecular entities (NME) and niche technology platforms, in-licensing them from or partnering with academic institutions and biotech or pharma companies, both locally and internationally. Projects are selected based on their novelty as a drug target or platform technology for diseases where there are unmet medical needs.
Upon reaching the clinical PoC, CALGENT will out-license or partner with the best-suited third party to continue development through market approvals and commercialization, maximizing profit through royalty, milestones, or profit sharing.

SoundJet Medical, Inc.
SoundJet Medical develops an innovative ultrasonic thrombolytic device for the treatments of pulmonary embolism (PE) and deep vein thrombosis (DVT), with a core technology named VorteSonic. VorteSonic uses ultrasonic tornadoes (ultrasonic vortex) to provide forces to loosen clots and penetrate thrombolytic drugs into clots simultaneously, resulting in very fast and complete thrombolysis. With VorteSonic technology, 40% of thrombolysis time is saved (in vitro data) compared with the solutions on the market, demonstrating the ability of less thrombolytic drugs within a treatment course, which further indicates reduction of hemorrhage risk and improved clinical outcome for patients.
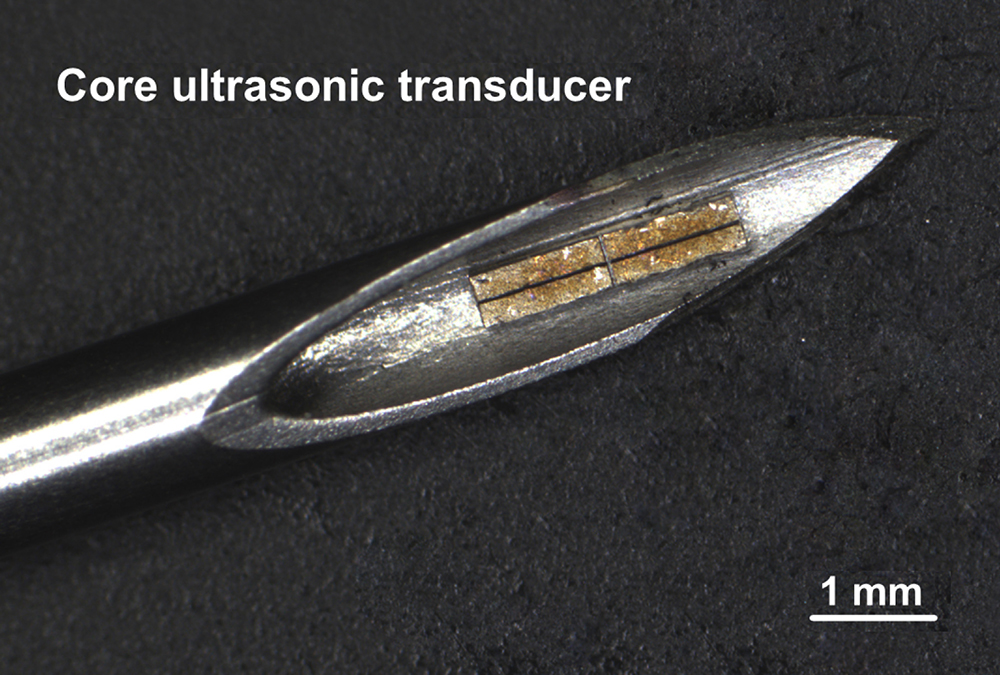
SoundJet Medical_core ultrasonic transducer
The core ultrasonic transducer generates ultrasonic tornadoes (ultrasonic vortex) to achieve fast and complete thrombolysis.
SoundJet Medical, Inc.
SoundJet Medical develops an innovative ultrasonic thrombolytic device for the treatments of pulmonary embolism (PE) and deep vein thrombosis (DVT), with a core technology named VorteSonic. VorteSonic uses ultrasonic tornadoes (ultrasonic vortex) to provide forces to loosen clots and penetrate thrombolytic drugs into clots simultaneously, resulting in very fast and complete thrombolysis. With VorteSonic technology, 40% of thrombolysis time is saved (in vitro data) compared with the solutions on the market, demonstrating the ability of less thrombolytic drugs within a treatment course, which further indicates reduction of hemorrhage risk and improved clinical outcome for patients.

Pharmasaga Co. Ltd.
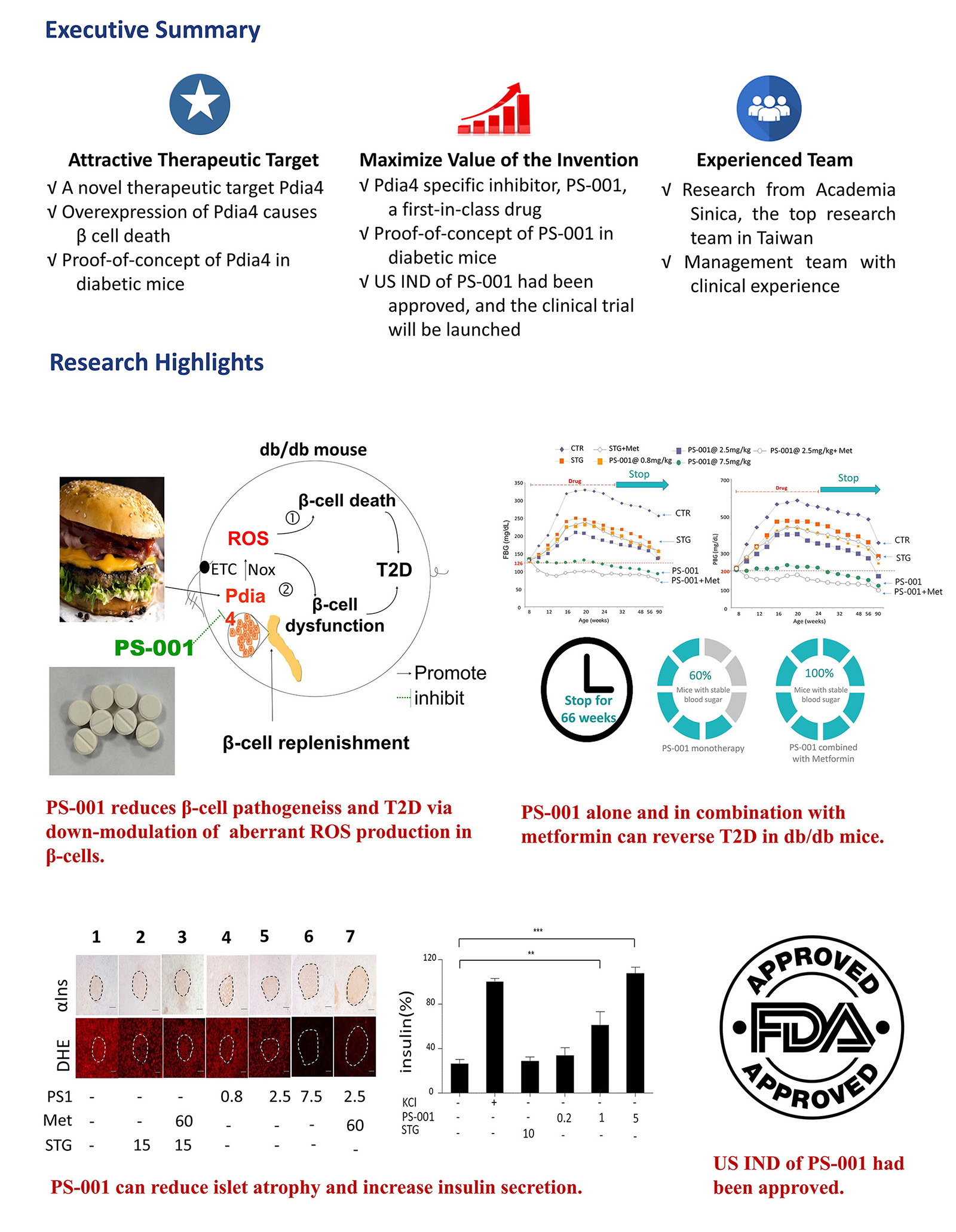
Pharmasaga is a drug discovery and development company located in Taiwan (http://www.pharmasaga.com/en/index.aspx). We aim to develop novel anti-diabetic small-molecule drugs that can treat and reverse diabetes.
Prevention of β-cell failure holds the key to curing diabetes. Pdia4 is a chaperone primarily expressing in β-cells. Genetics approaches showed that it promoted β-cell failure and diabetes in mouse models. Its novel mechanism involves regulation of oxidative stress in beta cells. Consequently, Pdia4 was identified as a new therapeutic target of diabetes (https://www.embopress.org/doi/full/10.15252/emmm.201911668). Using molecular docking, chemical synthesis and Pdia4 bioassays, we have finally identified drug candidates, e.g., PS-001 and PS-002. Both small-molecule drugs are Pdia4 inhibitors that can treat and reverse type 2 diabetes (T2D) and type 1 diabetes (T1D), respectively. For instance, PS-001 could inhibit β-cell failure and T2D via reduction of ROS generation. Moreover, 16-week treatment with PS-001 alone or in combination with metformin can reverse T2D in mice by 70% and 100%, respectively.
Our pipeline products include PS-001 for T2D and PS-002 for T1D. Of note, the IND of PS-001 was approved by US FDA, and its phase 1 clinical trial will start in late 2022, followed by phase 2 clinical trial starting in 2023.
Any pharmaceutical companies and investors that are interested in novel therapeutics for diabetes treatment and reversal are highly welcome.



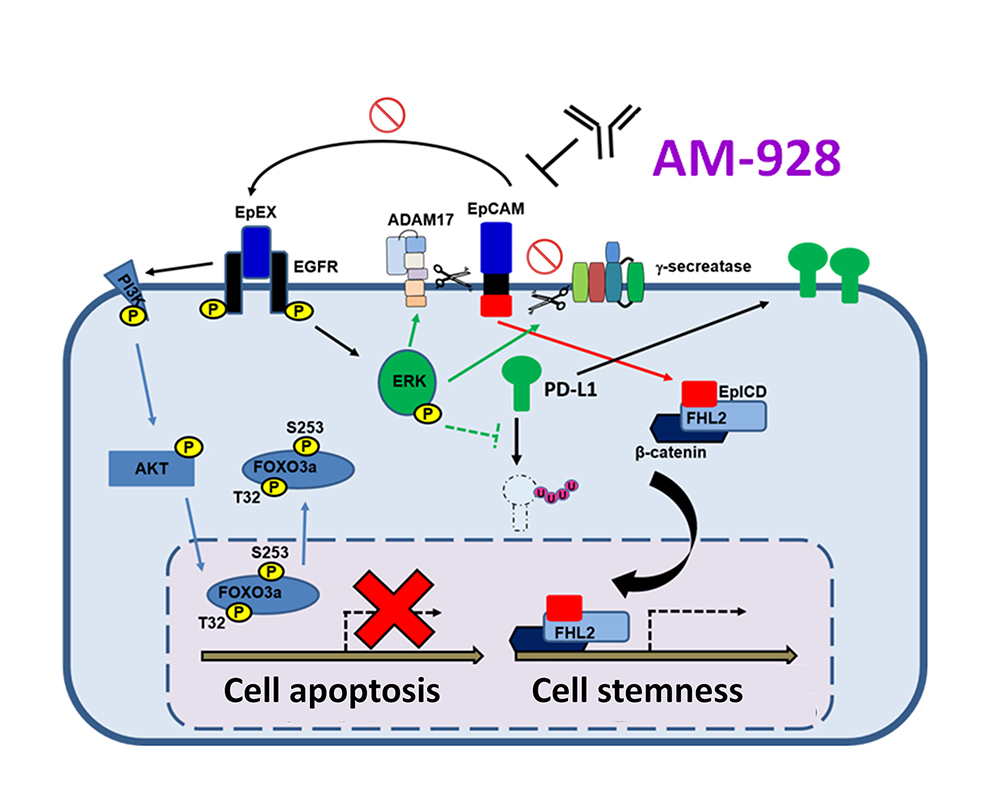
AM-928
AM-928 is a humanized antibody targeting epithelial cell adhesion molecule (EpCAM), which is highly expressed on the surface of solid tumors. The binding of AM-928 to EpCAM has potential for treatment of solid tumors by abolishing epithelial growth factor receptor (EGFR) signaling, inducing cancer cell apoptosis, and inhibiting tumor progression. AM-928 also possess the effector functions, such as antibody-dependent cell-mediated cytotoxicity (ADCC) for cancer killing.

PrecisemAb Biotech Co., Ltd.
We are PrecisemAb, a company that is committed to blocking side effects caused by antibody therapy. Our disease-directed Universal Antibody Lock is an autologous masking structure on the binding site of an antibody that allows antibody drugs to take effect only in diseased areas. This makes antibody treatment extraordinarily safe.
Compared to competitors, our Antibody Lock
1) is the 1st antibody lock that uses the antibody's own structure as a masking domain.
2) has higher masking ability and can fully activate the antibody when in the diseased areas.
3) has low immunogenicity and non-anti-idiotypic antibody binding, which prolongs serum half-life and leads to higher therapeutic efficacy.
4) takes only 3 months for Lock-design and function test.
We have successfully applied the technology to autoimmune diseases (TNFα and IL-1b) and immuno-oncology therapy (CTLA4, PD-1, 4-1BB). Potential application to ADC drugs is now under lead optimization. Through providing a customized lock-design service and co-developing lock-antibody drugs with pharma, PrecisemAb is dedicated to creating the safest antibody therapy for patients.
AHEAD Medicine
AHEAD Medicine unlocks the potential of flow cytometry and other high dimension data sources.
Increasingly precise lab equipment has begun to outstrip the capabilities of highly trained physicians and researchers, leading to delays, discordance, and excess costs. AHEAD uses machine learning and artificial intelligence to aggregate data across sites, provide deeper insights, and deliver faster and more accurate data interpretation all while lower testing costs.
Currently available for RUO, find out how we can help accelerate your research.
Calgent Biotechnology Co. Ltd.
Calgent Biotechnology Co. Ltd.
CALGENT, a spin-off company of Taipei Medical University (TMU), was established in 2019 and thrives on applying science and technology to drug development. CALGENT is committed to developing first-in-class drugs for novel therapeutic cancer targets and various treatments for ocular system, inflammatory and immune system related diseases.
One of the CALGENT’s main objectives is to develop exceptional and innovative medicines with the potential to bring positive and profound impacts on improving the life quality for patients with unmet needs.
Vision and Mission
CALGENT primarily focuses on the process from preclinical to clinical proof of concept (PoC) during the drug development value chain. CALGENT primarily targets highly novel projects with new molecular entities (NME) and niche technology platforms, in-licensing them from or partnering with academic institutions and biotech or pharma companies, both locally and internationally. Projects are selected based on their novelty as a drug target or platform technology for diseases where there are unmet medical needs.
Upon reaching the clinical PoC, CALGENT will out-license or partner with the best-suited third party to continue development through market approvals and commercialization, maximizing profit through royalty, milestones, or profit sharing.
Pharmasaga Co. Ltd.
Pharmasaga is a drug discovery and development company located in Taiwan (http://www.pharmasaga.com/en/index.aspx). We aim to develop novel anti-diabetic small-molecule drugs that can treat and reverse diabetes.
Prevention of β-cell failure holds the key to curing diabetes. Pdia4 is a chaperone primarily expressing in β-cells. Genetics approaches showed that it promoted β-cell failure and diabetes in mouse models. Its novel mechanism involves regulation of oxidative stress in beta cells. Consequently, Pdia4 was identified as a new therapeutic target of diabetes (https://www.embopress.org/doi/full/10.15252/emmm.201911668). Using molecular docking, chemical synthesis and Pdia4 bioassays, we have finally identified drug candidates, e.g., PS-001 and PS-002. Both small-molecule drugs are Pdia4 inhibitors that can treat and reverse type 2 diabetes (T2D) and type 1 diabetes (T1D), respectively. For instance, PS-001 could inhibit β-cell failure and T2D via reduction of ROS generation. Moreover, 16-week treatment with PS-001 alone or in combination with metformin can reverse T2D in mice by 70% and 100%, respectively.
Our pipeline products include PS-001 for T2D and PS-002 for T1D. Of note, the IND of PS-001 was approved by US FDA, and its phase 1 clinical trial will start in late 2022, followed by phase 2 clinical trial starting in 2023.
Any pharmaceutical companies and investors that are interested in novel therapeutics for diabetes treatment and reversal are highly welcome.

PrecisemAb Biotech Co., Ltd.
APrevent Medical
APrevent is a medical device company, founded by a team of international clinically experienced physicians and engineers. APrevent is dedicated to providing minimally invasive and innovative laryngeal implant and Software-as-a-Service (SaaS) technologies for voice & speech disorders, which are patient-orientated, physician friendly, and improve the quality of life for patients around the world. APrevent Intelligible speak, AiSpeak, is an innovative solution using big data and AI machine learning technology providing patients with motor speech disorders to communicate with their own speech, which recognizes and converts dysarthric speech into intelligible speech in real time. It can also diagnose and monitor conditions of a neurological disease based on the changes and variations of acoustic characteristics from patients. To help dysarthric patients regain normal communication capability and life quality, APrevent is developing several innovative solutions based on AiSpeak technology platform, including Portal and Messenger. AiSpeak opens a door to a complete new dimension of user experience for dysarthric patients with high potential value.

SoundJet Medical_control unit
The control unit performs as a coordinator for signal transmitting and receiving, as well as treatment control.

AcadeMab Biomedical Inc.,
FH Devices
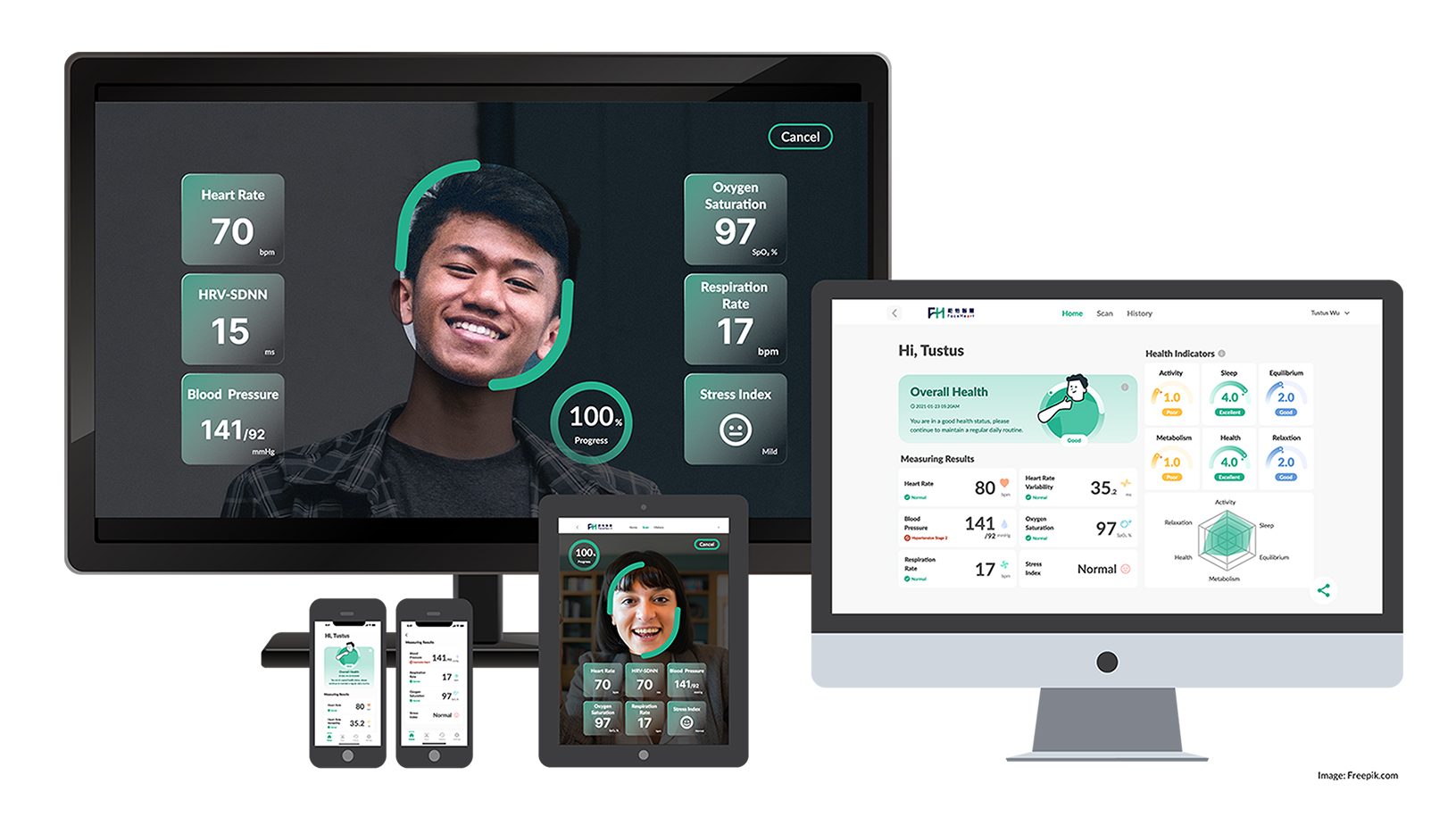
FaceHeart Vitals, connect AI from Face to Heart.
FaceHeart Vitals is an all-in-one vital monitoring solution for mobile health developers, healthcare professionals, and telemedicine practitioners. It is a patented Software Development Kit that utilizes the remote photoplethysmography (rPPG) method by incorporating a simple camera with image processing algorithms and AI technologies.
FaceHeart APP aims to improve human health via measuring six vital signs such as heart rate, heart rate variability, blood pressure, oxygen saturation, respiration rate, and stress index in only 40~60 seconds by integrating with various digital devices.
FaceHeart Reads Your Vitals Precisely, Through a Single-Camera Lens!
Calgent Biotechnology Co. Ltd.

Molsentech
Molsentech develops a 100% electrical-based and label-free disease detection platform powered by the Bio-FET technology that offers real-time and ultrahigh-sensitive detections.
Current detection platforms like PCR and ELISA are optical-based and there are complicated pretreatments needed to be done before the running real detections. Most of the time, their bulky equipment leads to high running and maintenance costs. To fix these pain points, we take advantage of the electrical-based Bio-FET technology that don’t require bio-labeling or amplification in pretreatments, so we can save a lot of time in the whole process. To be more specific, our analyzer can measure the immediate change in signals coming from the interactions between detection targets and bio probes we modify on the surface of biosensors to generate real-time results.
Our successful demo was COVID-19 RNA rapid test. Our testing time is as fast as antigen rapid tests, but we provide PCR-level accurate detections. Importantly, we just need about 3 months to develop a different biosensor assay for a new application and have started developing customized biosensors for biotech companies, which is our core B2B business model. This way, we could grow up by becoming a biosensor design house doing licensing and ODM so that we can expand our customer base and make a greater impact.
Our R&D members are from Academia Sinica and we have been on the BioFET research field for more than 2 decades. In addition to publishing papers with top SCI journals, in 2016, Molsentech signed a Joint-Development-Agreement with TSMC to develop Bio-FET biosensors together, the fact not only helped us save huge capital but also recognized our capability of facilitating this technology in the business world.
All in all, Molsentech has leveraged Taiwan’s semiconductor industry to make a promising technology ready to commercialize. Our vision is to become an important contributing factor to preventive healthcare & precise medicine. Molsentech is looking for strategic partners and investors, especially in the U.S., to successfully reach this goal with us.


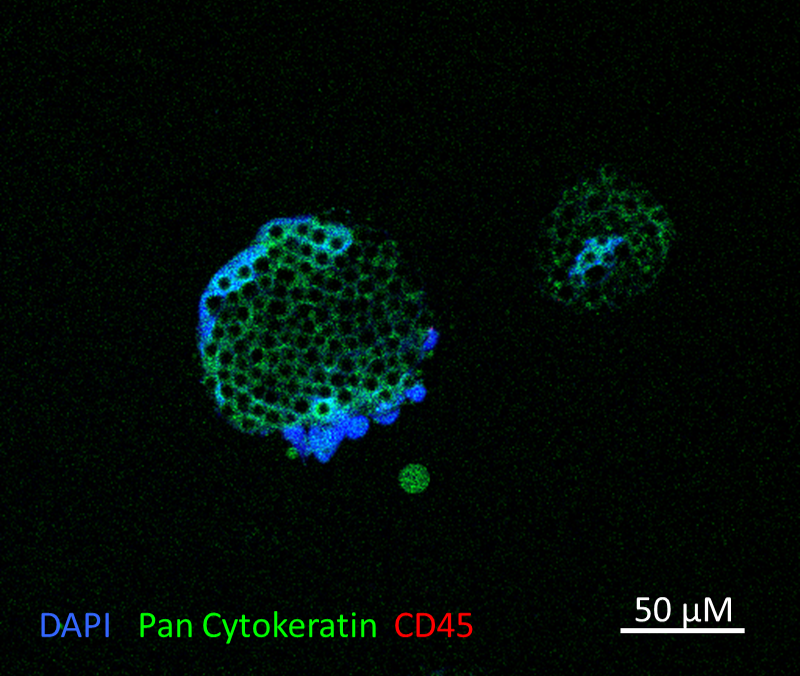
immnolfluorescence staining of CTC-derived organoid
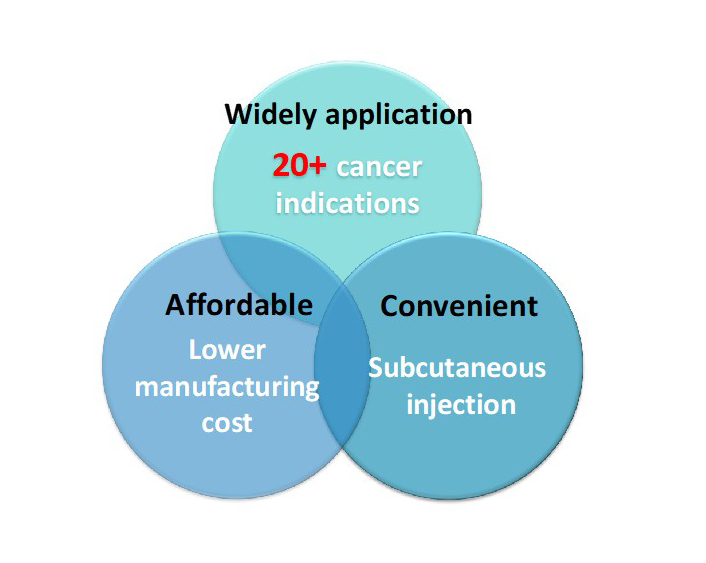
Advantages of therapeutic vaccine
We converted immune checkpoint drugs to therapeutic vaccine for animals that can be applied in more than 20 types of cancer, affordable and convenient injection.
CAL056
CAL056, a first-in-class, anti-cancer drug, modulates the proteasome pathway with low toxicity and a wide therapeutic window. Orphan drug designation was granted on Feb.28, 2019, and Phase I trial will be completed in Q3, 2022.
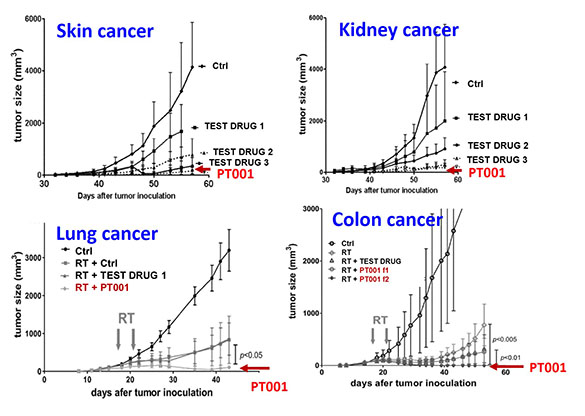
Preclinical results of therapeutic vaccine
Therapeutic vaccine (PT001) successfully inhibit tumor growth in mouse models.
Protect
Protect Biotech’s first broad-spectrum pet cancer immunotherapy therapeutic vaccine pipeline is ready to enter clinical trials. Our goal is to be a leader in promoting the health of companion pets
Anbogen Therapeutics
Anbogen Therapeutics is a clinical stage precision oncology company developing first/best-in-class personalized therapeutics for the treatment of solid tumors and blood cancers. We aim to bridge the gap between discovery candidates and proof-of-concept indications for unmet needs in clinical studies. We are advancing a pipeline of precision medicines for the treatment of solid tumors and blood cancers. Currently with three assets including ABT-101 and ABT-301 in clinical development.
Product
IA-05 is a truncated QS-21 analogue with improved tolerability, stability, and long-term immune responses in combination of various antigens. IA05 is expected to lead novel utilities in development of immunotherapies and therapeutic vaccines.
CancerFree Biotech Ltd.
CancerFree Biotech focuses on the use of Circulating Tumor Cells-Derived Organoid (CDO) culture system for a personalized analysis. We can rapidly amplify CTCs and develop an optimized anticancer strategy from the currently available anticancer drugs through low-risk and convenient liquid biopsy and biomimetic organoid culture system.

AcadeMab Biomedical Inc.,
AcadeMab Biomedical Inc., established in 2020, is a biotechnology company dedicated to discovering and early-stage development of proprietary biopharmaceuticals that applying our antibody development platforms. Our extensive practical experience with the platforms allows us to leverage resources and transform innovation that bringing up impact on disease treatment.
抗體鎖策略流程圖
Our disease-directed Universal Antibody Lock is an autologous masking structure on the binding site of an antibody that allows antibody drugs to take effect only in diseased areas. This makes antibody treatment extraordinarily safe.
Deep01 Limited
Deep01 is the developer of DeepCT - world-leading AI-powered brain CT abnormalities detection and quantification system. It detects brain bleeds, fractures, midline shift, and calculates ICH Score automatically.
Our AI makes doctors more efficiently in the medical process:
For Neurosurgeons - help to evaluate patients
For Emergency Physicians - help to triage patients
For Radiologists - help to generate reports

ABT-301
ABT 301, a small molecule drug with mechanism of action of histone deacetylase inhibitors (HDACi).

Awards

Protect
Protect Biotech’s first broad-spectrum pet cancer immunotherapy therapeutic vaccine pipeline is ready to enter clinical trials. Our goal is to be a leader in promoting the health of companion pets